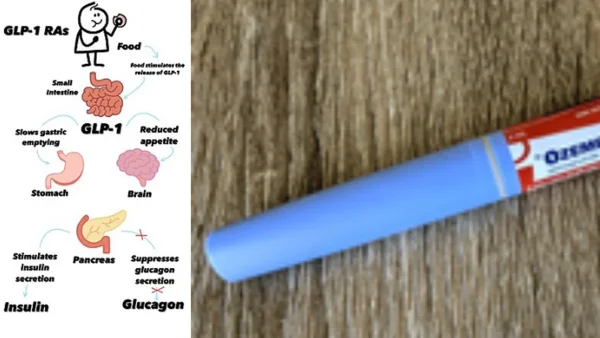
India has quickly increased how it’s checking on GLP-1 weight loss drugs, doing more inspections and enforcing the rules after all the new generic versions came out. The Drugs Controller of India, part of the Health Ministry, is now more carefully monitoring things to stop unauthorized sales, advertising the drugs for purposes they haven’re approved for, and people getting them on demand without a doctor.
What prompted the crackdown
The patent on semaglutide (the active ingredient in Ozempic and Wegovy) has expired, which means lots of generic versions are now being made. Over 40 companies are getting ready to make more than 50 different brands, and some are being sold for as little as 325 rupees a week.
Officials are concerned that having cheaper options so easily available will lead to misuse of the drugs.
Specifically, authorities are worried about the drugs being available throughout the whole system of getting medications to people, including at pharmacies, on websites, from wholesalers, and at wellness centers. They warn that using GLP-1 receptor agonists without a doctor watching over you can cause serious health problems and give people a false sense of security about quickly losing weight.
The Health Ministry has stated that making the drugs available and keeping people safe need to go together. Semaglutide and similar GLP-1 medications are only approved for certain medical conditions and need a trained doctor to start you on them, adjust your dose, and check on you regularly.
The enforcement playbook
Government regulators are working with each state’s drug authorities to check the entire pharmaceutical supply chain. In the last few weeks, they’ve looked at 49 different organizations in many areas, including warehouses for online pharmacies, drug wholesalers, pharmacies, and weight loss clinics.
These inspections were to find out about unauthorized sales, doctors not prescribing properly, and deceptive marketing. Organizations that weren’t following the rules have been given official warnings. Officials have said that they will increase monitoring in the next few weeks, and those who don’t comply will have their licenses taken away, be fined, and could be prosecuted in court.
Ban on surrogate advertising and off-label promotion
On March 10, 2026, the regulators told all the drug manufacturers they are not allowed to use “surrogate advertising” (hints or indirect advertising) or any other indirect advertising that could confuse customers or encourage them to use the drugs for something they aren’t meant for. They want advertising to stick to the approved uses of the drug and not blur the line between what is medically appropriate and what isn’t.
Who can prescribe GLP-1 therapies in India
In India, the drug is approved with limitations: only endocrinologists and internal medicine doctors can prescribe it, and in some cases cardiologists can too. The Health Ministry has told the public to only use GLP-1 weight loss drugs when a qualified doctor is supervising.
The most important thing is the safety of the patient. Using the drugs incorrectly, taking the wrong amount, or stopping them suddenly can be harmful. Doctors also say that the weight lost with these drugs can come back if you stop taking them, and so it’s important to make long-term changes to your lifestyle along with taking any medication.
Market dynamics and the public health backdrop
This move by the government is happening because of how bad metabolic health is in India. In 2021, approximately 101 million people had diabetes and 136 million had prediabetes, and around 315 million had high blood pressure. Obesity is increasing quickly, with about 180 million adults being overweight or obese, and that number is expected to increase sharply by the middle of the century.
More and more children are also being affected, which will cause problems for a long time to come. This explains why there’s a huge demand for GLP-1 drugs. However, experts are saying these are not quick fixes. They require a doctor to check you out, slowly increase the dose, keep track of any side effects, and combine the drug with changes to your diet, exercise, and behavior.
While many generic versions becoming available should make the drugs more affordable, lower prices could also mean more people will self-medicate inappropriately if things aren’t carefully managed. That’s why the government is doing two things at once: making the drugs available to patients who qualify and strongly controlling how they are sold and advertised.
What patients, providers, and platforms should do next
Before starting a GLP-1 treatment, patients should be seen by a qualified doctor, tell them about all of their medical conditions, and take the amount prescribed. They should also quickly report any side effects and avoid buying the medication without a valid prescription. Making lasting improvements to your lifestyle is still the most important thing for maintaining a healthy weight and metabolism.
Doctors and pharmacists should confirm what the medication is for, check for any situations where it shouldn’t be used, explain what to expect, and keep a record of follow-up appointments. Pharmacies and websites must be much better at checking prescriptions, prevent people from getting refills without a doctor’s approval, and remove anything that indirectly advertises the drug.
Regulators have said that enforcement will continue and be expanded. As more generic GLP-1 weight loss drugs become available, the message is clear: following ethical practices in the supply chain and having a doctor supervise is absolutely essential. Making the drugs accessible in a responsible way, and not making them available to anyone, will decide if these treatments will actually improve the health of the public in a large way.